Hydrochloric Acid
Muriatic acid
Structure
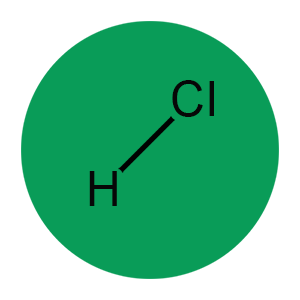
Formula: HCl (Cl(2)H)
Description: An aqueous solution of HCl. Colorless liquid with a pungent odor.
Uses: In the production of chlorides, refining ore in the production of tin and tantalum, for the neutralization of basic systems, as laboratory reagent, hydrolyzing of starch and proteins in the preparation of various food products, pickling and cleaning of metal products, as catalyst and solvent in organic synthesis, for oil- and gas-well treatment, in removing scale from boilers and heat-exchange equipment, pharmaceutic aid (acidifier).
Registry Numbers and Inventories.
| CAS | 7647-01-0 |
| EC (EINECS/ELINCS) | 231-595-7 |
| EC Index Number | 017-002-00-2 |
| EC Class | Toxic; Corrosive |
| EC Risk Phrase | R:34-37 |
| EC Safety Phrase | S:26-45 |
| ENCS (MITI) | 1-215 |
| RTECS | MW4025000 |
| RTECS class | Tumorigen (C); Mutagen (M); Reproductive Effector (T); Human Data (P); Primary Irritant (S) |
| UN (DOT) | 1789 |
| Merckn | 13,4801 |
| Listed on the Toxic Substancs Control Act (TSCA). | |
| Listed on Canadian Domestic Substances List (DSL). | |
| Listed on Australian Inventory of Chemical Substances (AICS). | |
Properties
| Formula mass | 36.46 |
| Melting point, °C | -114.9 |
| Boiling point, °C | -85.06 |
| Vapor pressure, mmHg | 88 torr (-115.5 °C) |
| Vapor density (air=1) | 1.25 |
| Critical temperature | 51.4 |
| Critical pressure | 81.6 atm |
| Density | 1.18 g/cm3 |
| Solubility in water | Miscible |
| Viscosity | 0.311 cp (-100 °C) |
| Heat of fusion | 1.98 kJ/mol |
| Heat of vaporization | 178 btu/lb |
| Odor threshold | >7.0 mg/m3 |
Hazards and Protection.
| Storage | Keep away from oxidizing agents, particularly nitric acid and chlorates. Safeguard containers against mechanical injury. |
| Handling | Containers of this material may be hazardous when emptied. Since emptied containers retain product residues (vapor, liquid, and/or solid), all hazard precautions given in the data sheet must be observed. Emergency eyewash fountains and safety showers should be available in the immediate vicinity of potential exposure. Do not puncture or incinerate containers. |
| Protection | Wear appropriate chemical protective clothing. |
| Respirators | Wear positive pressure self-contained breathing apparatus. |
| Small spills/leaks | Keep material out of water sources and sewers. Attempt to stop leak if without undue personnel hazard. Use water spray to knock-down vapors. Neutralize spilled material with crushed limestone, soda ash, or lime. Do not use water on material itself. Vapor knockdown water is corrosive or toxic and should be diked for containment. Land spill: Dig a pit, pond, lagoon, holding area to contain liquid or solid material. Dike surface flow using soil, sand bags, foamed polyurethane, or foamed concrete. |
| Stability | Stable – contact with common metals produces hydrogen which may form explosive mixtures with air. |
| Incompatibilities | Reacts rapidly and exothermically with bases of all kinds. Reacts exothermically with carbonates (and hydrogen carbonates to generate carbon dioxide Reacts with sulfides, carbides, borides, phosphides, many metals (including aluminum, zinc, calcium, magnesium, iron, tin and all of the alkali metals) to generate flammable hydrogen gas Reacts violently with acetic anhydride, 2-aminoethanol, ammonium hydroxide, calcium phosphide, chlorosulfonic acid, 1,1-difluoroethylene, ethylenediamine, ethyleneimine, oleum, perchloric acid, b-propiolactone, propylene oxide, silver perchlorate/carbon tetrachloride mixture, sodium hydroxide, uranium(IV) phosphide, vinyl acetate, calcium carbide, rubidium carbide, cesium acetylide, rubidium acetylide, magnesium boride, mercury(II) sulfate. |
| Decomposition | When heated to decomposition it emits toxic fumes of hydrogen chloride. |
| Other hazards | Corrodes many metals. Iron and aluminum are attacked readily. |

Fire.
| Flash Point,°C | 11 | |||
| Fire fighting | Extinguish fire using agent suitable for type of surrounding fire. (Material itself does not burn or burns with difficulty.) Cool all affected containers with flooding quantities of water. Apply water from as far a distance as possible. Use water spray to knock-down vapors. | |||
| Fire potential | May burn but does not ignite readily. | |||
| Hazards | Vapors from liquefied gas are initially heavier than air and spread along ground. Some of these materials may react violently with water. Containers may explode when heated. Ruptured cylinders may rocket. | |||
| Combustion products | Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. | |||
| NFPA | Health:3 Flammability:0 Reactivity:1 Special: – | |||
Health
| Exposure limit(s) | TLV: 5 ppm; 7.5 mg/m3 (ceiling values) (ACGIH 1992-1993). OSHA PEL: C 5 ppm (7 mg/m3) NIOSH REL: C 5 ppm (7 mg/m3) NIOSH IDLH: 50 ppm |
| Exposure effects | Shock, rapid breathing and pulse, circulatory collapse and other changes to pulse, blood pressure, and respirations may occur. Fetotoxicity, developmental abnormalities, and possible resistance to hydrogen chloride by inhalation during pregnancy have been noted. |
| Ingestion | Gastritis, burns, gastric hemorrhage, dilation, edema, necrosis, and strictures may occur. |
| Inhalation | Changes in breathing pattern, irritation, changes in pulmonary function, corrosion and edema of the respiratory tract, chronic bronchitis and noncardiogenic pulmonary edema have been observed. |
| Skin | Burns, ulceration, scarring, blanching, and irritation may occur. |
| First aid | |
| Ingestion | Have person drink water or milk; do NOT induce vomiting. |
| Inhalation | Remove person to fresh air; keep him warm and quiet and get medical attention immediately; start artificial respiration if breathing stops. |
| Skin | Remove and isolate contaminated clothing and shoes. Immediately flush with running water for at least 20 minutes. For minor skin contact, avoid spreading material on unaffected skin. |

Transport
| UN number | 1789 |
| Response guide | 157 |
| Hazard class | 8 |
| Packing Group | II; III |
| USCG CHRIS Codeh | HDC |
| USCG Compatatibility Group | 1 Non-oxidizing mineral acids |
